It appears Omaha Sen. Ernie Chambers has one less avenue to stop the Nebraska execution of condemned prisoner Carey Dean Moore in 15 days.��
After he challenged Pfizer pharmaceutical company Friday to use the courts to block the use of any of its restricted-use drugs in the possession of the Nebraska Department of Correctional Services, the company responded Monday.��
“Our records do not show any sales of any restricted products to the Nebraska Department of Corrections," said Pfizer spokesman Steven Danehy.
But just in case, the drug company asked the department again to return any Pfizer restricted products.��
The department has not responded to open-records requests by the Lincoln Journal Star, the Omaha World-Herald, other media and ACLU of Nebraska to reveal the sources of the drugs it plans to use in Moore's execution. Nor has it complied with a Nebraska district court judge's order to give the two newspapers and the ACLU certain records related to lethal injection drug suppliers.
People are also reading…
Another drug company, Alvogen, successfully filed a legal objection in Nevada this month to stop one of its drugs, the sedative midazolam, from being used in an execution. That execution did not take place as scheduled.��
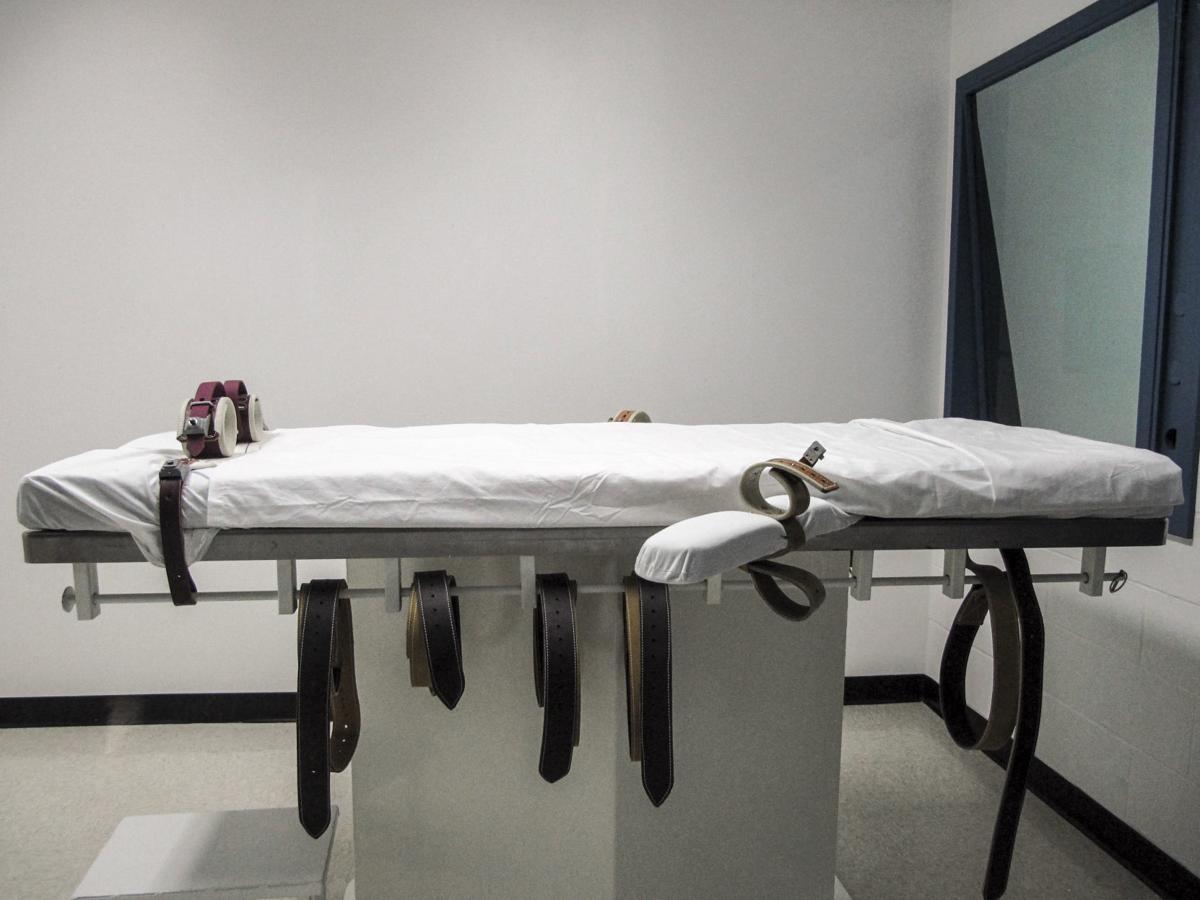
The Nebraska department has told Moore it plans to use four drugs, never before used in combination in an execution, in putting him to death. Those drugs are fentanyl, a powerful opiate painkiller; diazepam, which is the anxiety reliever Valium; cisatracurium besylate, a muscle relaxer; and potassium chloride, which stops the heart.��
Meanwhile, a second pharmaceutical company has joined a lawsuit to stop Nevada from using one of its drugs in the state’s execution of two-time murderer Scott Dozier.
According to the Las Vegas Review-Journal, a district judge Monday approved the July 24 motion filed by Hikma Pharmaceuticals, maker of the synthetic opiate fentanyl, to intervene in a lawsuit previously filled by Alvogen.��
Also, a "friend of the court" brief was filed with the U.S. Supreme Court on July 23 by the Association for Accessible Medicines, which represents generic drug manufacturers, in connection with a challenge by a death row inmate in Missouri.
The brief argued that manufacturers overwhelmingly oppose the use of their products for lethal injections, and that no prescription drug has been tested or approved by regulators at the high doses typically employed in an execution protocol. Nor is lethal injection a medically accepted off-label use of the powerful injectable drugs used as part of execution protocols, the brief said.